Whales Present Specific Challenges for Evolution
Nearly all mammals are tetrapods, having both a front pair and a hind pair of limbs. They generally have a mouth full of hard teeth, and have nostrils near the front of their snout.
Whales are clearly mammals: they nourish their unborn young through a placenta, feed newborns with milk, and so on. However, whales are thoroughly adapted to living in the water. Modern whales and dolphins typically display no hind legs, their nostrils are located near the top of their heads, and they have various features in their head and jaws that facilitate hearing underwater. Also, one major class of whales lacks regular teeth: the “baleen” whales use porous baleen plates to filter out food from the water.
If evolution with common ancestry is true, today’s whales have developed from earlier mammals with hind legs and teeth, and nostrils in the front of their snouts, and we should find physical evidence of that ancestry, in their genomes and in the fossil record. Evolution thus makes some well-defined hard predictions, which can allow us to disprove it if it is not true. This is the way science progresses. If the predictions of a theory are found to be valid over and over again, across a wide variety of circumstances, we become more justified in holding to this theory. If the physical predictions are found to fail, we must either modify the theory or abandon it.
Evolution demands that many intermediate species existed between modern whales (with no hindlegs and with nasal openings atop their heads) and their earliest, purely terrestrial ancestors (with four legs and nostrils at the front of their snout). These species would have physical features lying between these early ancestors and modern whales, and thus would exhibit shrunken hindlegs, and nostrils partway back on their snouts. While acknowledging that we are not able to recover fossils of every single species that ever lived, we expect from evolution to find at least a few such intermediate specimens in the ancient rock layers.
The ancestors of whales possessed genes for producing hind legs and teeth. Thus, from evolution we expect to find these genes for hind legs in the genomes of whales, even though those genes are not expressed in actual hind limbs. Similarly, we would expect to find the genes for fully enameled teeth in the genomes of the baleen whales, even though those teeth are not actually formed in the adult whales. Let’s look at the evidence, and see whether it supports these specific predictions from evolution. I have tried to provide enough details here for a fair assessment, but that has made for a rather long article. Feel free to scroll down to the CONCLUSIONS section for the final results.
CONTENTS
Whales Present Specific Challenges for Evolution
What Is a Whale?
GENETIC EVIDENCE FOR WHALE EVOLUTION
Are There Genes for Hind Legs in Today’s Whale Genomes?
Are There Genes for Teeth in Today’s Toothless Whale Genomes?
Other Genetic Evidence for Common Descent
WHALE EVOLUTION FOSSIL INTERMEDIATES
The Nature of the Fossil Record
Whale Fossil Nostril Position Shifts with Time
Changes in Hind Legs and Hips
Distinctive Ankle Bones Are Consistent With Genetic Link to Hippopotamus
SOME OBJECTIONS FROM OPPONENTS OF EVOLUTION
CONCLUSIONS
FURTHER RESOURCES
ENDNOTES
~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~
What Is a Whale?
Here is a simplified classification of “cetaceans”:
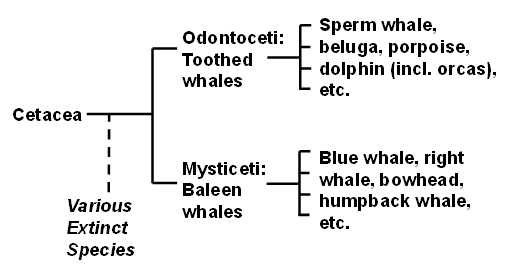
The word “whale” is used in two main senses. In the larger sense, it can refer to cetaceans as a whole. This would include the all the various Odontoceti (toothed whales, including dolphins) and the Mysticeti (baleen whales), as well as the various extinct whales found in the fossil record. This is the sense in which we will generally use the word here. Sometimes, though, “whale” is used to denote the cetaceans other than dolphins and porpoises.
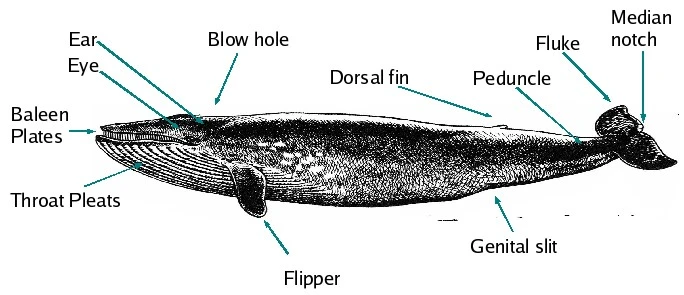
Features of a Blue Whale, from https://en.wikipedia.org/wiki/Whale Image: Jim Thomas
Shown above is a figure of a blue whale. The blue whale is the largest animal ever known to exist, with a maximum recorded weight of 190 U.S. tons (173 tonnes). This figure shows some features shared by all whales, along with other features that are distinctive to only certain types of whales. First, note the complete absence of hind legs. Also, instead of two nostrils at the front of the snout, there is a pair of “blow-holes” at the top of the head. Whales (unlike fish) have lungs and need to take breaths of air to live, so having the blowhole at the top of the head makes it easier for the whale to breathe while in a horizontal position just below the surface of the water.
The blue whale is a mysticete (baleen) whale. Its baleen plates are shown in the figure. Mysticeti force seawater through the bristles of the baleen, in order to trap smaller fish, invertebrates, or plankton, which the whale then eats.
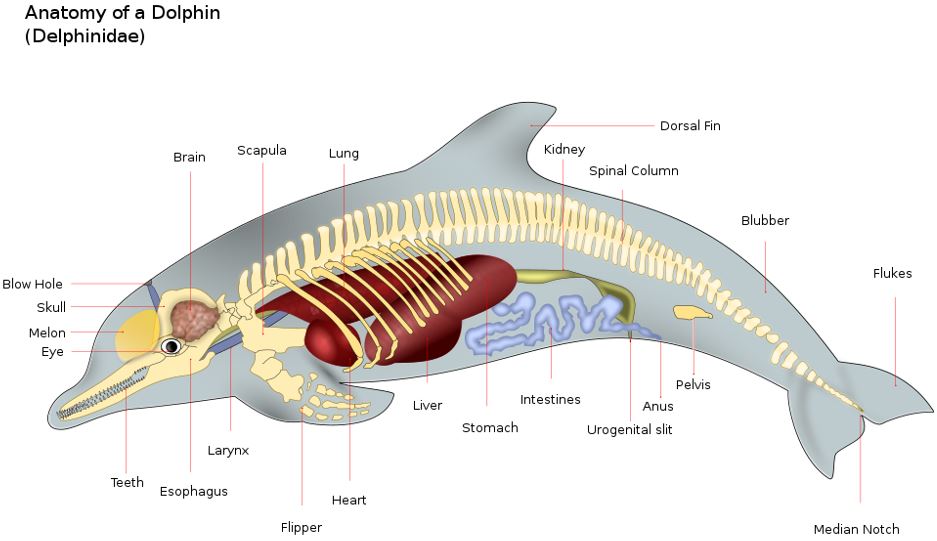
Dolphin Anatomy, from https://en.wikipedia.org/wiki/Cetacea , image version by The Photographer
The anatomy of a dolphin (a toothed whale) is shown above. Again, the blowhole is at the top of the head. As a predator, the dolphin has teeth. It also uses an echo system to locate prey. It generates clicks in its nasal passages, and these clicks are focused forward via the “melon” organ in the forehead. Like other whales, the dolphin hears sounds (including the return echoed clicks) primarily via the lower jawbone, which conducts sounds to the ear. We will discuss more details of ear anatomy below.
The Distinctive Ear Feature of Cetaceans
A distinctive skeletal feature that appears in all cetaceans, but in no other living animal, is a thickened wall of the tympanic bone in the middle ear. This ear bone is shaped like a half walnut shell, with an empty cavity in the middle. In nearly all mammals, the thickness of the wall of this cavity is more or less constant all around the ear. In whales, however, the internal wall is much thicker than the external wall. This thickened wall is called the involucrum. This distinguishing feature is also found in a series of fossils with whale-like features, going back for tens of millions of years. Scientists have decided to classify all such creatures as “cetaceans” or whales, even though the further back in time we go, the less these fossil animals look like today’s whales. Some of these fossil animals (e.g. Pakicetus, Indohyus) have legs instead of flippers, and clearly lived on land, although their features suggest that they spent much time submerged in rivers. Living whales, and most fossil whale intermediates, also have a wavy crest on the tympanic plate, on the opposite wall of the tympanic bone from the involucrum. This wavy edge is called a sigmoidal process. This sigmoidal process is present in Pakicetus, although there it is more plate-like than the sigmoidal process in the dolphin ear, which features a finger-like projection.

Tympanic ear bone for fossil Pakicetus and for modern dolphin, showing thickened involucrum of inner wall. From The Walking Whales (2014), by J. G. H. “Hans” Thewissen
Another example of the involucrum and sigmoid process of a fossil whale earbone is shown below.

Features of a fossil whale earbone, including thickened involucrum and sigmoid process. From definition of “Sigmoid Process” in the Paleos vertebrate glossary.
~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~
GENETIC EVIDENCE FOR WHALE EVOLUTION
Are There Genes for Hind Legs in Today’s Whale Genomes?
It is common to find some deactivated genes in organisms. These are recognizable as having the DNA sequences for generating proteins, except for some mutation(s) in the gene or in its regulatory region(s) that prevent the original protein from being made. In some cases, this same gene is found in other species, where it does in fact cause a protein to be made. These defunctionalized genes are called pseudogenes. In the course of evolution, some of the genetic material in pseudogenes may get repurposed. Thus, while they no longer code for their original protein, many pseudogenes have acquired some other functionality in the genome. It is not correct to refer to them all as “junk”.
A well-known example of a pseudogene in the human genome is the “GULO” gene. Mammals have a set of genes, including one called GULO, for synthesizing vitamin C (ascorbic acid) which is an essential chemical for metabolism. In some mammals, such as guinea pigs and many primates (including humans), there are mutations in the GULO gene which render it ineffective for making vitamin C. Therefore, humans and these other species must now consume vitamin C in their foods to stay healthy, since (unlike most mammals) they cannot synthesize it themselves. There is reason to believe that the ancestors of today’s primates (and guinea pigs) ate a diet rich in vitamin C, so the GULO gene function was not as important to survival; hence deactivating mutations in the GULO gene could accumulate in the species without killing it off. Retaining a deactivated GULO gene in the human genome is predicted by evolution, so finding it there furnishes further evidence of common ancestry.
As noted, common descent demands the presence of genes for growing hind limbs in the ancestors of today’s whales. Barring some improbable genetic deletion event, we expect to find these genes still in the genomes of current whales, even though they are no longer serving their original function.
Studies with other mammals have identified a number of genes which are critical in the formation of hind legs. These include the “sonic hedgehog” (SHH) gene, along with genes FGH8 and FGH10, all of which have been identified in whale genomes. The gene which is perhaps most specifically associated with hind limb formation is known as TBX4. When we peer into a whale genome, there it is:
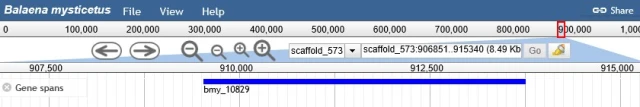
Location of TBTX4 gene in bowhead whale genome, here labeled bmy_10829. From http://www.bowhead-whale.org/annotations/details/bmy_10829/
Thus, as predicted by evolution, the genes for making hind legs are still present in whales. We now know as a result of additional study that these genes have additional functions in embryonic development, so they need to be present in whales even if they do not produce hind legs. But this does not detract from the value of the initial prediction of their presence based on common ancestry.
Even before the advent of whole genome sequencing for whales, it could be inferred that most of the genetic machinery for growing hind legs existed in the whale genome. Observations showed that cetacean embryos actually do grow small hind limb buds, which do not progress to full limbs (see Figure 2.4.2 in TalkOrigins article ). A 2006 study by Thewissen et al. tracked the development of dolphin embryos in detail, identifying which genes were activated (or not) for the hind limbs at various points in embryonic development. Although all the necessary genes are still present, there are changes in their expression which keep the hind limbs from growing past the bud stage in the embryo. Thus, the loss of hind-limbs in whales (like many other seemingly large evolutionary changes) did not require the production of novel genes, but instead involved more subtle changes in the regulation of existing genes.
Occasional mutant cetaceans have been observed in which the hind-limbs are more fully developed, as in the dolphin below. This again demonstrates that the genetic machinery for growing hind-limbs is still present in whale genomes, even though it is not normally expressed:

From TalkOrigins : The hind-flippers of a bottlenose dolphin found in Japanese coastal waters. Yellow arrows indicate the location of a well-formed atavistic set of hind limbs. (Images modified from publicity photos from the Taiji Whaling Museum.)
A whaling ship off the coast of British Columbia captured a humpback whale in 1919 which had two hind-legs protruding from it. The skeletal remains of one of the legs, which was analyzed by an expert from the American Museum of Natural History in New York. The legs were initially over four feet long, and covered with blubber and skin. More details on this find, including photographs, are given at TalkOrigins.
Are There Genes for Teeth in Today’s Toothless Whale Genomes?
As noted above, common ancestry and evolution predict that the baleen whales should retain the genes for making enameled teeth, even though such teeth do not actually grow in their mouths. Dennis Venema at Biologos has assembled the key facts, and (in italics) we quote here extensively from his article “Whale Evolution: Theory, Prediction and Converging Lines of Evidence“:
Some of the genes known to be used in all mammals for tooth formation were the obvious candidate genes to start with: the products of the ameloblastin, amelogenin, and enamelin genes are all used in the formation of tooth enamel, the hardest structure in the vertebrate skeleton. Researchers went looking for these genes in several Mysticete (i.e. toothless whale) species. The results showed that all the species studied did indeed have these three genes present as pseudogenes.
In other words, these genes were present (each with crippling mutations), which was what evolution predicted. The figure below shows part of the sequence of the ameloblastin (AMBN) pseudogene in the DNA of a baleen whale, compared to functional AMBN genes from a toothed whale and from other species. The baleen whale genome has suffered the insertion of an extra base pair (shown here as a “C”), which will throw off the reading frame for part of the gene. This kind of frameshift mutation can dramatically alter the expression of a gene, typically making it useless for producing the original protein. Except for that inserted “C”, the sequence shown here for the toothed whale is nearly identical to that for the hippopotamus. In general, the hippo genome is more similar to whales than it is to any other extant mammal.

Caption: Alignment of ameloblastin (AMBN) enamel gene sequences (F) from a baleen whale, a toothed whale (“Tooth”), and more distantly related mammals with single base-pair frameshift insertion in the baleen whale AMBN pseudogene (pink). Source: McGowan, et al., Trends in Ecology & Evolution, June 2014, Vol. 29, pp. 336-346.
However, as biologists examined the details of these mutations, they were troubled by the patterns they found. Data from the fossil record suggested that teeth were lost only once, and early in the lineage leading to all modern toothless whales. That led to the expectation that all the modern toothless whales would share fairly similar disabling mutations in these genes, or in at least one of them. However, that was not the pattern they found: …none of the mutations that removed the functions of these three genes were shared between different species, suggesting that these genes lost their function independently in the species studied.
Reasoning within the framework of evolution led to a further proposal, that a fourth gene necessary for enamel formation was lost early in the evolution of the mysticetes (baleen whales):
The loss of any one gene necessary for forming enamel would be enough to prevent the process altogether. In this case, the loss of this fourth gene would prevent tooth enamel from forming, even though the genetic sequences of the other three enamel genes would still be intact. Once enamel function was lost, random mutations in the remaining enamel genes could then accumulate later in Mysticete evolution after speciation in this group was already underway.
Guided by this prediction, researchers examined other, related genes and found the expected gene which had the same deactivating mutation across all modern toothless whales. Quoting again from the Venema article:
The smoking gun for tooth loss in Mysticetes turned out to be exactly what was predicted: a fourth gene, necessary for enamel production, and mutated with the same inactivating mutation in all modern toothless whales. The gene in question, named enamelysin, was destroyed when a mobile genetic element called a SINE transposon inserted into it, breaking it into two halves and removing its function:

The fact that the same SINE insertion mutation at an identical location is found in all modern Mysticete species indicates that this mutation happened once in a common ancestor and then was inherited by the entire group. Since this must have occurred early in the evolution of toothless whales in order to happen in the common ancestor of the entire group, the picture from the genetics and the fossil record match. Once again, findings in one discipline (in this case, paleontology) can be used to make very detailed predictions about what another, unrelated discipline (comparative genomics) should reveal. These results are also entirely consistent with the observation, made in the 1920s, that toothless whales form tooth buds during embryogenesis that are later reabsorbed prior to the point when the deposition of enamel would begin.
[ Text and Figure from D. Venema, Whale Evolution: Theory, Prediction and Converging Lines of Evidence ]
The only reasonable explanation for the presence of these disrupted tooth enamel genes is that they were originally functional genes in some ancestral species, but that they underwent significant mutations in the course of evolution. The other possibility is that God created these mysticete species with these nonfunctional genes in them, to make it look like evolution is true. But that is not a “reasonable” explanation, since God (in the Bible, anyway) is associated with truth, not deception.
Further Genetic Evidence for Common Descent
Deactivated Olfactory Receptor (OR) and other genes
Olfactory receptors (ORs) are proteins which are expressed in the cell membranes of olfactory receptor neurons. They bind to various compounds with have odors, which gives rise to the sense of smell. Olfactory receptor neurons in mammals connect to the olfactory bulb of the brain.
It is not clear how well the ORs which aquatic whales inherited from their terrestrial ancestors (for smelling in air) continue to function under water. Odontoceti (toothed whales) locate prey primarily by echolocation, rather than smell. Their nasal passages have been remodeled to facilitate emitting pulses of sound; they lack an olfactory bulb and an olfactory nerve.
We therefore expect from evolution that the genomes of these toothed whales would still have many genes for making ORs left over from their terrestrial ancestry, but (since the need for the function of these proteins has largely disappeared), that a high percentage of these genes would have suffered disabling mutations. That is indeed the case. The figure below shows that for the seven toothed whale species listed, the percent of recognizable OR genes that have been disabled from their protein expression function (i.e. have become pseudogenes) ranges from 74% to 100%. The percent of OR pseudogenes in Mysticeti (baleen whales) is much lower (35% for the species shown here), i.e. the fraction of functional OR genes is higher. The baleen whales retain an olfactory bulb and olfactory nerve, and probably do utilize some sense of underwater smell.
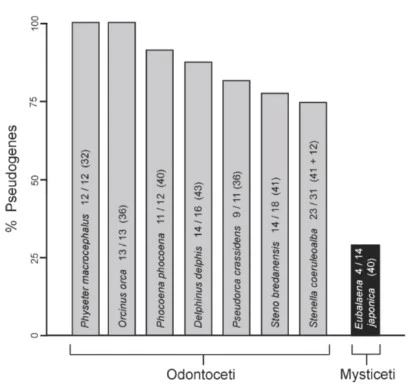
Percent Pseudogenes among Olfactory Receptor (OR) genes, for various whale species. Screenshot from https://biologos.org/resources/the-tale-of-a-whale-an-ideal-case-study-for-introducing-the-evidence-for-evolution , where this figure is credited to McGowen, et al., 2008.
McGowen, et al. published in 2014 a massive study of differences in whale genes compared to terrestrial mammals, which indicate how whales came to have the physical features which they do. Some figures from that paper are reproduced below.

Above is the color key for figures below, showing modifications in genes or gene expression in whales, or at least some whales, compared to other mammals. The figure below shows the anatomic location for expression of the genes in question. For instance, the Olfactory Receptor (OR) genes, associated with the nasal area, are shown in red since they have been largely inactivated in some whales. A number of other types of genes in the whale genome have also been inactivated, including other genes for taste and smell (e.g. TAS1R and V1R in toothed whales), some retinal genes (e.g. OPN1SW), and genes for tooth enamel (in baleen whales), as discussed earlier. As discussed earlier, the SHH and HAND2 genes involved in making hindlimbs are still functional, but have been greatly down-regulated, hence they are coded purple, per the color key above.
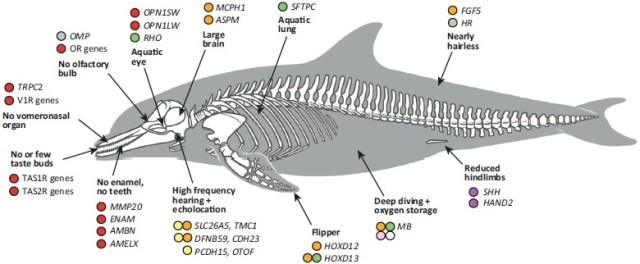
Some differences in genes/expression between whales and terrestrial animals, mapped onto anatomy. Figure from Michael R. McGowen, John Gatesy, Derek E. Wildman, “Molecular evolution tracks macroevolutionary transitions in Cetacea”. Trends in Ecology & Evolution, Volume 29, Issue 6, June 2014, Pages 336-346. [As with all images here, click it to see clearer image]
Below is shown a lineage diagram among major living whale groups, and non-whales (“Outgroups”), depicting points at the various changes in gene expression have occurred. Again, red dots indicate inactivated genes, and so on. The pattern here is a nested hierarchy, which is strong evidence for common ancestry via evolution. The caption for the original figure reads in part:
Major molecular changes in Cetacea mapped onto a phylogenetic hypothesis (A) and to corresponding anatomical features (B). Colored circles identify the type of molecular change. Single asterisks show that the molecular change occurred within the given clade; for example, inactivation of OPN1LW* on the Physeteroidea branch indicates that pseudogenization occurred within Physeteroidea (in this case, independently in Physeter and in Kogia). Double asterisks mark genetic changes that have been inferred based on the bottlenose dolphin genome, and are mapped arbitrarily to the Delphinoidea branch. Inferred positive selection in MCPH1, ASPM, MB, HOXD12, and HOXD13 is mapped to the stem cetacean branch and indicates general evidence for positive selection at these loci in Cetacea.

Some differences in genes/expression between whales and terrestrial animals, mapped onto major whale classes. Figure from: Michael R. McGowen, John Gatesy, Derek E. Wildman, “Molecular evolution tracks macroevolutionary transitions in Cetacea”. Trends in Ecology & Evolution, Volume 29, Issue 6, June 2014, Pages 336-346.
Deactivated genes for clotting and other functions
Progress continues to be made in understanding how various genetic mutations have contributed to the adaptation of whales to a marine environment. A 2019 study by Huelsmann, et al. provided additional details on how the deactivation or down-regulation of specific genes in whales can explain many of their unique body features that are so well adapted to living in the ocean and diving deeply. The research team compared whale genomes to that of hippopotamus (the closest living terrestrial genetic relative of whales) and other mammals to identify novel gene-deactivating mutations that are shared by both odontocetes (toothed whales) and mysticetes (baleen whales). After excluding members of the olfactory receptor and keratin-associated gene families, whose losses have been already been studied in detail by previous researchers, and after applying other screening, they identified a set of 85 broken genes that exhibit shared inactivating mutations in odontocetes and mysticetes.
After further study of the functions of the intact versions of these genes in other mammals, the researchers identified eight genes whose deactivation can be tied to specific beneficial biochemical alterations in the whales. As one example, two of these genes, F12 and KLKB1, promote clot formation. When whales dive, they are at risk for forming unwanted clots, due to constriction of blood vessels and the appearance of tiny nitrogen bubbles in the blood. These bubbles can serve as surfaces which trigger clotting.
The deactivation of F12 and KLKB1 reduce the risk of harmful clotting. Some specific spots in these genes where deactivating mutations are shown in the figure below. Compared to other mammals, whales have a two-base insertion (“GG”) in F12, which throws off the transcription of the gene. For KLKB1 a whole chunk containing exons 6-12 has been deleted. There are additional disabling mutations in both genes. Again, the most reasonable explanation for the presence of these disabled genes is that they are inherited from ancestral species, but were significantly modified in the course of evolution.

Examples of disabling mutations for genes F12 and KLKB1 in whales (red font). These genes in other mammals promote blood clotting (thrombosis). Source: Huelsmann et al., Sci. Adv. 2019; 5 : eaaw6671 25 September 2019.
WHALE EVOLUTION FOSSIL INTERMEDIATES
The Nature of the Fossil Record
It is typically observed that a species appears suddenly in the fossil record without a clear, graduated set of intermediate forms between it and some previous species. This is precisely what we expect from the following two facts:
( 1 ) Very, very few of all the organisms that have died in past eons became fossilized, and only a small fraction of those that did fossilize have been found. As we can observe today, nearly all carcasses rot or are eaten by scavengers rather than being buried intact in rock layers and forming hard, detectable fossils. Of the remains that do get fossilized, many are later eroded away, or smeared beyond recognition in metamorphic transformations deep in the earth. Also, of all the potential fossil-bearing rocks that survive today, only a small fraction of them are available near the surface for paleontologists to examine.
The Coelacanth fishes furnish a classic example of the fickleness of the fossil record. The Coelacanth order of fishes was once widespread in the ancient seas. Coelacanths peaked in the fossil record about 240 million years ago, and then declined. The most recent known fossil dates back to about 80 million years ago. It was thought that they had become extinct. In 1938, however, a live coelacanth was discovered in the Indian Ocean. Since then a number of others have been caught. (As might be expected, these modern specimens are not precisely the same species or even genus as the fossil coelacanths, but they are clearly coelacanths). Unless we are prepared to claim that an Intelligent Agent supernaturally re-created these modern coelacanths, we must acknowledge that some population of these fish has existed for the past 80 million years but without leaving a trace in the fossil record.
(2) The basic arithmetic of population genetics shows that it is difficult for new genetic mutations to become established in very large populations. Thus, it is far more likely that a new species will develop within a small, isolated population, especially if that population is under some environmental stress that would favor genetic changes. Such a small, transient population is unlikely to leave a trace in the fossil record. If the new species becomes more fit than the old species, the new species will expand in numbers and only then is likely to appear in the fossil record. But once a species is widespread and successful in its ecological niche, there will be diminished selection pressure for changes, so fossils of this now well-adapted species may appear for millions of years with showing little change.
Evolutionary lineages tend to be “branchy”. Typically the organisms out on the side branches show up in the fossil record, rather than the transitional onesalong the main “trunk” of the evolutional family tree. The transitional ones along the “trunk” would have existed in small, isolated populations whereas some of the organisms on the side branches will be the large, established, stable populations, which will leave appreciable fossils. This trend is illustrated below.
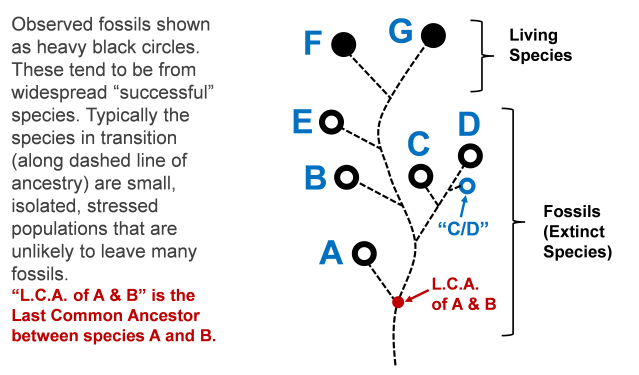
Figure: Representative lineage, based on available fossil evidence at the “tips” of the lineage tree branches.
In this figure, living species are shown as solid black circles, and fossil (extinct) species as heavy black circles. As noted, the species that actually leave appreciable amounts of fossil evidence will tend to be large, stable populations, whereas the small, isolated, possibly stressed populations in transition will likely not leave enough fossils to be found by us millions of years later.
Thus, we should expect many gaps in the observable fossil record. The fact that various transitional fossils have not yet been found is not a rational basis for believing that these transitional forms never existed. It is worth noting that as time goes by, more and more gaps do get filled in by additional fossil discoveries (as predicted from common descent). However, there will always be some gaps left. For instance, in the figure above, if a fossil “C/D” is found in rock layers between C and D, and with features intermediate between C and D, that would still leave gaps (though smaller gaps) between C and C/D, and between C/D and D. Opponents of evolution will continue to complain about these gaps, no matter how small they become.
In the popular literature, species A might be referred to as the “ancestor” of B and the other species shown. However, this is imprecise and generally incorrect. The other species are not direct descendants of A. The actual last common ancestor between A and the rest is shown as “L.C.A. of A & B”. Using the discipline of cladistics, we can often make reasonable estimates as to what this common ancestor looked like and when it existed, but there is always some uncertainty about its features and dating. The actual common ancestor (for all the reasons discussed) is unlikely to have left enough of a fossil trace to be observed by us today.
Also, the exact lineal relations among the fossil species cannot be determined with 100% certainty. In the figure above, the dashed lines could reasonably be drawn somewhat differently than shown. For instance, perhaps “C” should be shown as branching off from the line leading to “B” rather than from the line leading to “D”. Scientists are guided by patterns of various details in the fossil remains (cladistics) in proposing where the ancestral splits in lineage occur, but these proposals are always subject to revision. Thus, if some estimate of ancestral relations at the lower species/genus/family level is revised in the light of further evidence, that is standard, responsible science in action. It does not constitute grounds for rejecting common ancestry as a whole.
Despite these inherent uncertainties in the details, the fossils that HAVE been found fit well within this conceptual framework. Opponents of evolution like to trot out outdated, out-of-context quotes from scientists which seem to admit to a dearth of transitional fossils. The reality is: “So many intermediate forms have been discovered between fish and amphibians, between amphibians and reptiles, between reptiles and mammals, and along the primate lines of descent that it often is difficult to identify categorically when the transition occurs from one to another particular species.” [Science, Evolution, and Creationism by the National Academy of Sciences]
There is a clear, general progression of life-forms found in the fossil record. Trilobites (early arthropods, distant cousins to today’s insects and crustaceans) always appear in sedimentary layers below any rocks containing dinosaurs (reptiles), never above. In the vertebrate lineage, first fish, then tetrapod amphibians, then reptiles, and finally mammals make their appearances in the rock layers. A number of fossils with part fish/part amphibian characteristics, and with part reptile/part mammal characteristics, are found at about the times expected for these transitions. This is all consistent with common ancestry within the theory of evolution.
When we move from the analysis of broad groupings of organisms (classes, orders, etc.) and instead zoom into the details the appearance of individual species, however, the fossil record appears “jumpy” rather than smoothly continuous. Anti-evolutionists claim that this disproves evolution. However, as explained above, this pattern is consistent with the facts that (1) the fossil record is known to be extremely incomplete, and (2) transitional forms are most likely to arise in small populations. Thus, it is quite reasonable to hold that transitional species did exist, but not in sufficient amounts or locales to leave discoverable fossils.
When proposed lineages versus geological time are portrayed in journals, they are often shown with right angles and horizontal and vertical lines. It is understood that in reality the evolutionary changes between fossil species would occur gradually, i.e. with sloped or curving lines of bodily changes versus time, not in sudden vertical jumps. However, since we generally don’t have enough information to depict the exact evolutionary trajectories of the intermediate species, the lines are drawn at right angles simply to make the proposed ancestral relations as clear as possible for the fossils we do have. The figure below shows the extended cetacean lineage. The closest living relative to whales is the hippopotamus. This style of drawing lineages also makes it clear that the species or groups shown, which are known from the fossil record, are regarded in general as residing on side branches from the lineages of the actual transitional species. Again, the species shown are not considered to be direct ancestors; cetaceans did not descend from hippopotami, but from some (typically unknown) common ancestor. For a more thorough presentation of these general fossil concepts, and detailed examples of transitional fossils, see Realistic Expectations for Transitional Fossils .

Cetacean Lineage. From https://en.wikipedia.org/wiki/Whale
Whale Fossil Nostril Position Shifts with Time
As noted above, evolution and common ancestry demands that many intermediate species existed between extant whales and their earliest, purely terrestrial ancestors. These species would have physical features lying between these early ancestors and modern whales. Fossils typically preserve bones, but not soft tissues. Thus, we will focus here on skeletal features such as hind legs, nostrils, and ankles. Other special adaptations of whales, such as how the young are born and suckled, heat exchange, hemoglobin, etc., as not as easy to track through fossils, and will not be addressed here.
One of the earliest fossil species determined to be in the whale ancestral line is Pakicetus, dated about 52 mya (million years ago). (We noted above the distinctive earbone feature that defines cetaceans). Like most terrestrial mammals, its nasal openings are near the front of the snout. In contrast, the nostrils of modern whales lie far back on their heads, essentially on top of their heads, roughly above the eyes. The modern whale nasal openings are sometimes called blow-holes.
We expect from evolution that, with successive generations, the nostrils moved in stages from the front of the snout for Pakicetus, backwards to the top of the head for modern whales. While we can never find all the fossils of all the intermediates, we would expect from evolution to find at least some fossil species, dated between 52 mya and the present, with nostrils positioned between the front and the top of the head. And that is exactly what we do find.
The figures below depicts the nasal openings (black) for a chronological series of five toothed whales on the left of the blue line, running from 52 mya to the present. A general progress of the nostrils to a more rearward position is evident. To the left of the blue line is the skull of a fossil Aetiocetus (23-24 mya, with mysticete features) with the nasal opening halfway back, compared to a modern mysticete (baleen whale, Balaenoptera) with nostrils further back.
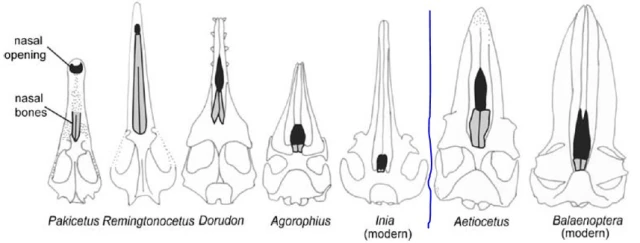
Figure showing nasal bones (gray) and nasal opening (black) in skulls of some fossil and modern whales. The five species to the left of the vertical blue line are all toothed whales, in left-to-right chronological order (c. 52 mya for Pakicetus, c. 43-45 mya for Remingtonocetus, c. 34-40 mya for Dorudon, c. 32 mya for Agorophius). The two skulls to the right of the blue line are baleen whales, again in left-to-right chronological order (c. 23-24 mya for Aetiocetus). Source: Thewissen, et al., Evo Edu Outreach (2009) 2:272–288.
Here is another set of whale skulls, two fossil (47 mya and 36 mya) and modern. These again show a general shift backwards in nostril position with time. For the middle skull, the nostrils lie roughly halfway back on the head.
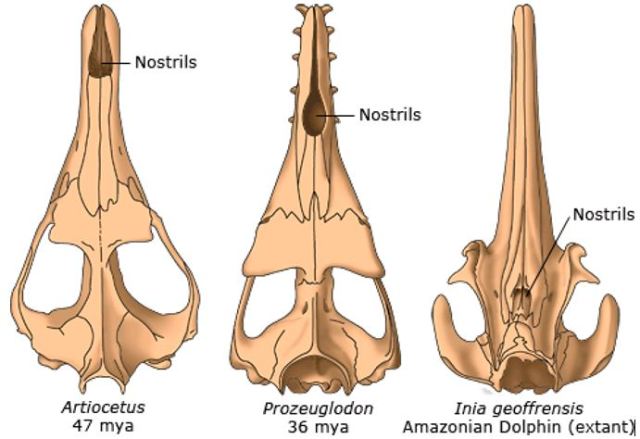
Three whale skulls, showing change in position of nostrils with time. From Berkeley The evolution of whales, https://evolution.berkeley.edu/evolibrary/article/evograms_03.
Evolution does not always progress in the same direction and at the same pace in every branch of a lineage. Also, a species or genus or family may persist for millions of years with little apparent change. Thus, we would not expect that if every known fossil whale were lined up in chronological order, that there would be a perfectly smooth, uniform repositioning of the nostrils. Nevertheless, there is a general trend in nostril position from the front of the snout for the very earliest, fully terrestrial species (c. 45-52 mya), to middle positions for many species between about 25 and 45 mya, to the top-of-the-head position for all modern cetaceans.
Changes in Hind Legs and Hips
Modern whales (apart from very rare mutants) have no hind legs. As we look backwards in time, we expect from evolution to find some fossils with small, vestigial legs, and then larger, more functional legs on the earliest terrestrial ancestors.
First, we look back to some fossils from the 35-40 mya timeframe, Basilosaurus and Dorudon. These whales look very much like modern whales in overall appearance, but they do have little hind legs. These legs have all the hallmarks of being vestigial appendages. They are far too small to function as weight-bearing legs, but they retain many of the physical features of regular terrestrial legs. Basilosaurus legs have a femur (thigh bone), a knee joint with patella (kneecap), and a complete ankle with tibia and fibula. A Basilosaurus ankle is shown below, with lower tibia and fibula, and several toebones:

Source: West Virginia University Geology 331 class notes
The pelvis of the Dorudon (below) is unattached to the spine, and thus would not bear any appreciable weight. However, again, the hind leg structure mimics terrestrial legs, with thigh (femur) and ankle bones, and toes.
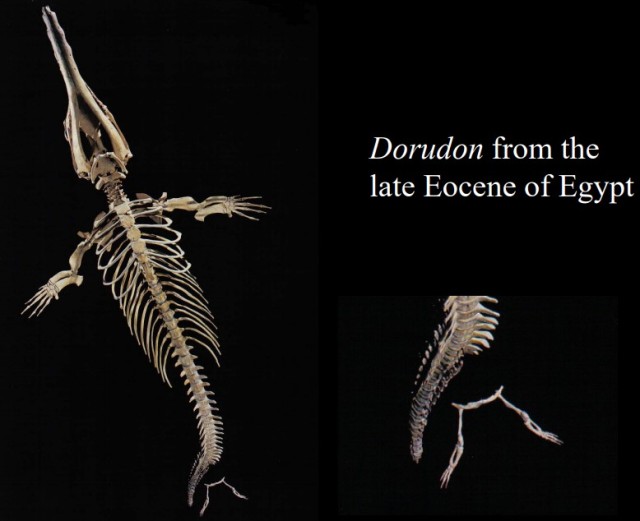
Source: West Virginia University Geology 331 class notes
This figure compares the skeletons of Basilosaurus and Dorudon with Pakicetus and another intermediate species, Ambulocetus. Several lines of evidence indicate that Pakicetus, while fully terrestrial, did go into rivers to eat fish. Ambulocetus was more adapted to aquatic life, with proportionately shorter legs and large feet, which facilitated paddling although they made Ambulocetus more unwieldy on land. Most of the bones for Ambulocetus have been found, but not the snout, so we do not know exactly where its nostrils were situated; the picture below is a reasonable reconstruction based on the known bones and comparisons with similar fossil species.
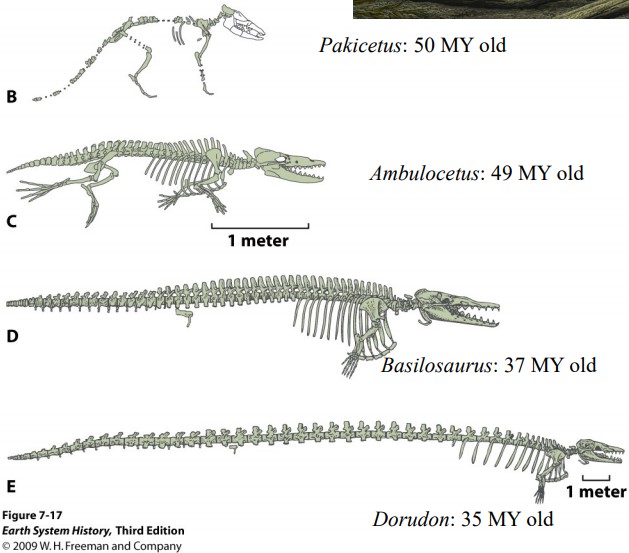
Source: West Virginia University Geology 331 class notes,
The figure below includes the skeleton of Rodhocetus, which was even more adapted to marine life than Ambulocetus.

Source: https://thewhalesevolution.weebly.com/the-whale-evolutionary-tree.html
The legs of Rodhocetus are shorter, and its tail stronger, than for Ambulocetus. Features on the toe bones indicate the feet were webbed, and the toes terminated in nails or small hoofs. Its footbones were so long and thin that they would not support the animal’s weight on land, so when Rodhocetus came up onto the land, it would have to walk essentially on its heels. The large webbed feet would, however, be effective for paddling in water. The tail was strong and powerfully muscled, to aid in swimming. Rodhocetus fossils have been found in locations indicating it swam far out to sea, although it apparently fed mainly in near-shore locales. The hips were attached to the spine, allowing the hind legs to bear some weight, but the pelvis is attached to fewer vertebrae compared to typical terrestrial mammals. Also, the vertebrae of the sacrum (the portion of the vertebral column that is connected to the pelvis) are not fused together, again differing from typical land mammals. The unfused sacrum allows the vertebral column to be more flexible during swimming, although giving less support to the body on land.
An article by Robert Boessenecker of the New York Institute of Technology has more details about Rodhocetus, including some whale-like adaptations for underwater hearing:
The lower jaw has a large hole near the jaw joint (the mandibular foramen), which in modern toothed whales, is filled with fat (“the mandibular fat pad”). In living odontocetes, sound travels through the thin bone on the side of the jaw and is channeled into the middle and inner ear by the pad of fat. In land mammals (including humans), underwater hearing takes place by sound waves traveling through the bones of the skull to the ears. Sound traveling in this manner arrives at each ear at nearly the same time, and it is not possible to tell the direction from which the sound came. The fat pad in the lower jaw of Rodhocetus and some other cetaceans allows directional hearing when underwater. This indicates that Rodhocetus was well adapted to the aquatic environment….. The teeth in the snout (incisors and canines) are cone-shaped (like many modern fish-eating toothed whales), suggesting that Rodhocetus fed upon fish. … Carbon isotope analysis of the teeth of Rodhocetus suggests that it fed in areas of low salinity (such as estuaries and other shallow marine environments), in contrast to the deep ocean environment of deposition for the sedimentary rocks that entombed it.
Here are the bones recovered for an early (1994) partial specimen of Rodhocetus; note the nostril opening is back from the tip of the snout, but not as far back as the basilosaurids:
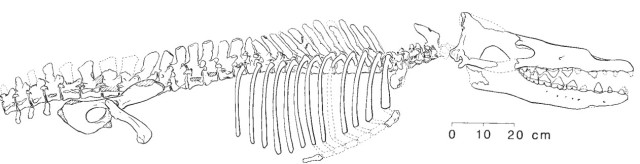
Source: Gingerich, et al., Nature, vol. 368, p. 844 (1994).
An artist’s conception of the appearance of Rodhocetus, based on more complete fossil evidence, is shown below:

Source: Cover of Science (2001) Philip D. Gingerich (umich.edu)
Some of the stepwise changes in physical features we have discussed in the course of whale evolution are shown in the figure below:

Source: The evolution of whales , https://evolution.berkeley.edu/evolibrary/article/evograms_03
The next figure also delineates the progression of physical changes, in more detail. The oldest species are at the bottom of the diagram. The step by step by step changes observed in the fossil record, occurring steadily over millions of years, are powerful confirmation of the evolutionary origins of today’s whales. The original figure caption reads:
Phylogenetic relationships of Cetacea based on paleontological and molecular evidence showing the sequential acquisition of derived aquatic specializations in the group. Internodes for outgroup branches (A,B), the stem lineage of Cetacea (C–M), the stem lineage of Mysticeti (N–S), and the stem lineage of Odontoceti (T–V) are
colored and labeled with capital letters. Major character transitions and/or acquisitions are listed to the right and are ordered by internode (A–V) from the base of the tree.
For character mappings that are ambiguous, a range of nodes is given. Approximate branch lengths are from [reference] [5], and brackets to the right delimit groups. Fossil lineages are dashed, and extinct genera are marked by crosses. Paintings are by Carl Buell.

Source: Michael R. McGowen, John Gatesy, Derek E. Wildman, “Molecular evolution tracks macroevolutionary transitions in Cetacea”. Trends in Ecology & Evolution, Volume 29, Issue 6, June 2014, Pages 336-346.
Here we expand the lower half of the figure above, to better display the transitional changes for the earliest cetacean species. A total of eight intermediate fossil cetaceans are shown in the series from terrestrial Pakicetus to totally aquatic Basilosaurus :

Source:Michael R. McGowen, John Gatesy, Derek E. Wildman, “Molecular evolution tracks macroevolutionary transitions in Cetacea”. Trends in Ecology & Evolution, Volume 29, Issue 6, June 2014, Pages 336-346. [Again, click image for clearer view]
Rodhocetus, while well-adapted for aquatic life, still had hind legs functional enough to allow some locomotion (however clumsy) on land. Basilosaurus and Dorudon are totally aquatic – – their hind legs are tiny, and their hips are not attached to their spines, and so cannot bear any weight on land.
While this set of species is already an impressive transitional series, it would be nice to have even finer morphological gradations to fill in the (relatively small) gap between Rodhocetus and Dorudon. Was there any fossil species which had more substantial hind legs than Dorudon but could not effectively walk on land? As it turns out, such an animal has been found, namely Georgiacetus.
Georgiacetus, shown in the diagram above, was closer in form to modern whales than was Rodhocetus. Several partial skeletons are available. The first specimens (encased in rock) were dug up in 1983. After years of painstaking removal of rock from around the bones, the first analysis was published in 1998. The known Georgiacetus fossils (c. 41 mya) are about five million years later than Rodhocetus, and slightly earlier than the basilosaurids (i.e. Basilosaurus and Dorudon, c. 34-40 mya). The pelvis of Georgiacetus was proportionately much larger and with better-developed hip sockets than for basilosaurids, indicating substantial hind legs for paddling. However, its pelvis (as with Basilosaurus and Dorudon) was not attached to its spine, so it could not support its weight walking on land. Like later whales, none of its vertebrae were fused into a sacrum. Accordingly, its whole spine could participate in undulation for swimming. Its fossils have only been found in marine sediments, and analysis of its bones indicates it drank saltwater, not fresh water. Therefore it probably lived full-time in the ocean, like modern whales. Georgiacetus is thus a significant intermediate between Rodhocetus and the basilosaurids. See an article by Jonathan Geisler of the New York Institute of Technology for more details.
Wikipedia commented on the similarities between the skulls and teeth of Georgiacetus and the basilosaurids:
“The tooth and skull arrangement shows a clear, direct relationship between the Basilosauridae family and Georgiacetus. Basilosaurids possessed teeth and a skull remarkably similar in function and structure, both have nostrils (blowholes) located halfway back on the snout, just in front of the eyes. The basilosaurid tooth arrangement appears as a more efficient and specialized version of the Georgiacetus arrangement. Georgiacetus predated the basilosaurids by nearly three million years; the Georgia Whale was very likely the source of the basilosaurids. In their turn, the basilosaurids led to modern whales.”
Aegicetus fossils were discovered in the Egyptian desert in 2007, and are dated about 35 mya, which is contemporary with basilosaurids (c. 40-34 mya). Aegicetus shares many features with Georgiacetus, such as legs smaller than Rodhocetus yet proportionately larger than basilosaurids, and unfused sacral vertebrate with no attachment of the pelvis. Again, this would preclude effective walking on land.
Distinctive Ankle Bones Are Consistent With Genetic Link to Hippopotamus
The closest genetic relative to today’s whales is the hippopotamus. This relation was controversial when it was first discovered, because whales are carnivores, whereas hippos are primarily herbivores. In the twentieth century, an extinct group of carnivores called mesonychids was widely proposed as terrestrial ancestors for whales. They seemed to be the closest relatives to whales, based on the fossil record at that time. Thus, in the late 1990’s there seemed to be a discrepancy between the fossil record (which favored mesonychids) and the genetic evidence which favored a lineage shared with hippopotamus. Although anti-evolutionists sometimes still cite papers from that era to claim a contradiction between the fossils and the genetics, in fact this controversy was resolved with further fossil finds around 2000, which clearly supported the link with hippopotamus as opposed to mesonychids.
Hippopotamus is a type of artiodactyl. Artiodactyls are hoofed animals (ungulates) with an even number of weight-bearing toes. A distinctive skeletal feature of all living artiodactyls is an ankle bone (astragalus or talus) with double cusps on the upper and lower surfaces. This is referred to as a “double pulley” shape. We might therefore expect from the genetic data that the earliest antecedents to whales would likewise possess an ankle bone of this shape. And they do.
Below is a figure showing the astragalus of a Pakicetus, demonstrating how similar it is to modern artiodactyls such as pig and deer, and different from other animals such as dogs. Again, Pakicetus is a very early (c. 52 million years ago) animal which was identified as a cetacean by its ear structure. This anklebone shows consistency with the comparative genetic data.

This image is from Campbell 11th edition, Figure 22.19, p 475. (Credit: Thewissen lab), and was taken from a Biologos presentation.
As already noted, Rodhocetus was a later (c. 46-47 mya) species which was more adapted to water than Pakicetus and thus more like modern whales. It, too, displays ankle bones similar to modern artiodactyls. The slide below compares the ankle of Rodhocetus with that of a hippo. Again, Rodhocetus shares the distinctive “double pulley” anklebone, consistent with the common ancestry of all whales (fossil and living) with hippos.

Fossil whale intermediate Rodhocetus ankle, compared to hippo ankle. Red arrow points to distinctive “double pulley” anklebone, found only in artiodactyls like hippos and in fossil whale intermediates. Screen shot from YouTube video What is the Evidence for Evolution? Credit: Philip D. Gingerich, U. Michigan.
In the picture below are shown the central ankle bones (astragali) of three artiodactyls (two early fossil whale ancestors with robust legs, and a modern pronghorn antelope). They have double pulley joints and hooked processes pointing up toward the leg-bones. On the right is a photo of the tiny vestigial hind foot of a basilosaurid, dating about ten million years later than the other two fossil whales. As noted earlier, Basilosaurus has a complete ankle and several toe bones, even though it can’t walk. The basilosaurid astragalus still has a pulley and a hooked knob pointing up towards the leg bones as in artiodactyls, while other bones in the ankle and foot are fused. The Berkeley commentary on this figure notes, “From the ear bones to the ankle bones, whales belong with the hippos and other artiodactyls.”

At left, the ankle bones of two middle Eocene protocetid archaeocetes, Rodhocetus balochistanensis (left) and Artiocetus clavis (right) from Pakistan (both around 47 mya), compared to those of the modern pronghorn Antilocapra americana (center). At right, the ankle region and foot of Basilosaurus (c. 37 mya). The upper pulley part of the astragalus (outlined) connects to the tibia and fibula. From Berkeley The evolution of whales, https://evolution.berkeley.edu/evolibrary/article/evograms_03
SOME OBJECTIONS FROM OPPONENTS OF EVOLUTION
Who Are the Opponents of Evolution?
The vast majority of those in North America who oppose evolution are conservative Christians. These fall into two main groups. One group is the Young Earth (YE) creationists, who hold the earth was created in six 24-hour days, about 6000 years ago. They take their stand on the Bible as their primary source of understanding the past, which trumps any possible physical evidence to the contrary. Unfortunately, they elevate their interpretation of the Bible to the same sacred level as the Bible itself.
They are typically ignorant of the history of how Genesis has been interpreted in the past. For instance, for most of the twentieth century, almost no conservative Protestants believed that the earth was only 6000 years old. Most fundamentalists held to the “gap” theory of interpreting Genesis 1:1-2, whereby the initial creation of the earth was perhaps billions of years ago, but the final reshaping of the earth and the creation of modern animals and humans occurred more recently. W.B. Riley, editor of The Christian Fundamentalist and president of the Anti-Evolution League of America, stated in the 1920’s that there was not “an intelligent fundamentalist who claims that the earth was made six thousand years ago; and the Bible never taught any such thing”. Other Protestants had other ways of understanding Genesis which also allowed for an old earth. Roman Catholics typically have no problem with an old earth.
The Bible often presents spiritual or moral teachings in the form of stories or imagery which are not literally true. It is true that the simplest, most literal readings of Genesis 1-3 and other passages point to a recent creation. However, it is also true that the simple, literal meanings of many Biblical passages show that the earth is stationary, and the sun and other celestial objects revolve around the earth. These verses include Psalm 104:5 (“He set the earth on its foundations; it can never be moved”), Ps. 93:1 (“Surely the world is established, so that it cannot be moved”), I Chron. 16:30 (“The world also is firmly established, It shall not be moved”), the philosophical discourse of Eccl.1:5 (“The sun also rises, and the sun goes down, and hastens to the place where it arose”), and also the historical chronicle of Josh. 10:13 (“So the sun stood still in the midst of heaven, and did not hasten to go down for about a whole day”).
In the 1500s and 1600s, the literal interpretation of these passages was seen as an essential element of Christian belief. John Calvin stated that people who claim that the earth “shifts and turns” were monstrous, malicious, and devil-possessed. Cardinal Roberto Bellarmine, a prosecutor of Galileo, stated in 1615: “…to affirm that the sun is really fixed in the center of the heavens and the earth revolves swiftly around the sun is a dangerous thing, not only irritating the theologians and philosophers, but injuring our holy faith and making the sacred scripture false.” Note well: “…injuring our holy faith and making the sacred scripture false.” That is what today’s YE creationists say about an old earth and evolution, i.e. that these concepts injure our faith and make the sacred scripture false.
What has changed since 1600? Once theologians finally accepted what the scientists had been telling them about these aspects of the physical world, the theologians took a fresh look at the issue and found that, lo and behold, a literal acceptance of a stationary earth was not essential to the Christian faith after all. If we accept that the Bible was written originally to an ancient Near Eastern people, with their ancient physical conception of the world, it can be seen that the Genesis creation narrative (which is consistent with ancient science) was a marvelous means of communicating key concepts like monotheism and the dignity of mankind to that people at that time. I have sketched out some principles here for interpreting Genesis in a way which honors God’s revelation in his works (i.e. the physical world) as well as in his Word.
It was only the publication of The Genesis Flood in 1961 which popularized the young earth position. As described here, the “Flood geology” on which this book is based was inspired by a nineteenth century cult prophetess, Ellen White. Flood geology is not based on actual science, and indeed it is soundly refuted by a simple look at common rock formations. YE creationism denies much of modern science, including astronomy, geology, and biology.
Nevertheless, modern YE creationists are convinced of their position, and often denounce Christians who do not agree with them as “compromisers”. As stated in the preface to The Genesis Flood (6th printing), “We take this revealed framework of history [i.e. recent six 24-hour day creation] as our basic datum, and then try to see how all the pertinent data can be understood in this context”. In other words, their literalistic interpretation is non-negotiable, and they determine to fit the physical evidence within this framework. If any facts contradict their interpretation, they feel obligated to subvert or simply ignore these facts.
Intelligent Design (ID) proponents constitute the other main group of evolution opposition. They are a more diverse grouping than YE creationists. There are exceptions, but the majority of ID proponents are evangelical Christians. In general, they accept nearly all of modern science, including the Big Bang origin of the universe and the commonly accepted ages of the fossils.
They are troubled, however, by the possible atheistic implications of evolution via plain natural processes, which they mistakenly equate with “materialism”. The epicenter of the ID movement is the Discovery Institute in Seattle. A founding document of this organization reads:
Thinkers such as Charles Darwin, Karl Marx, and Sigmund Freud portrayed humans not as moral and spiritual beings, but as animals or machines who inhabited a universe ruled by purely impersonal forces and whose behavior and very thoughts were dictated by the unbending forces of biology, chemistry, and environment… The cultural consequences of this triumph of materialism were devastating… Discovery Institute’s Center for the Renewal of Science and Culture seeks nothing less than the overthrow of materialism and its cultural legacies.
ID proponents have tried to present themselves as objective “detectors of design”, but their underlying religious and philosophical motivations cannot be disguised. Although they stoutly deny it, in practice the methodology of ID proponents is nothing more than the old “God of the gaps” gambit. That is, they try to identify things that modern science cannot (yet) completely explain, and use those to claim that science is hopelessly confused, and that therefore we need to invoke the intervention of a superhuman “Intelligent Designer” to account for these things. Unfortunately, in their zeal to discredit modern science, they misrepresent the actual state of affairs.
Whereas YE creationism has a positive agenda (i.e. a clearly-stated Flood geology model which purports to account for the observed state of the earth), ID is purely negative. While sniping at current scientific models, ID proponents refuse to propose an actual testable model of their own. [1] Any possible physical finding can be viewed as, “Well, that is just the way the inscrutable Intelligent Designer chose to do it.” Thus, ID does not qualify as any kind of science. While being untestable shields ID from being disproven, it also means there cannot possibly be any evidence which positively supports ID.
For instance, if an Intelligent Designer did intervene to cause the appearance of a long series of animals in the 35-50 mya timeframe which were more and more adapted to marine life (e.g. Ambulocetus and Rodhocetus, then Georgiacetus, then Dorudon, and so on), how exactly did he/it intervene to make this happen? Did he miraculously create one species after another, every million years or so, in an order which just happened to resemble what unaided evolution would produce? Or did he reach in and supernaturally tweak generations of genomes to somehow give a subtle assist to evolution via common descent? Or what??? ID proponents refuse to say.
There is a tragic irony in ID proponents viewing evolution according to the ordinary regularities of nature as being synonymous with “materialism” and thus being incompatible with their Christian faith. Instead of “overthrowing” materialism, they are making materialism more plausible. This fear of evolution is bad philosophy and bad theology, showing a lack of understanding of the sovereignty and providence of God. If I can believe that God was providentially involved in how I obtained the particular genome I did from my parents, a process involving mutations and rearrangements according to ordinary physical operations (no supernatural intervention), then I can acknowledge God’s same providential involvement in the billions of mutations and rearrangements of genomes (again, with no necessary supernatural interventions by an Intelligent Agent) that led from fish to me.
By virtue of its assumptions and methodology, science is very good at doing one thing, which is explicating regularities of events in the physical world. It has nothing whatsoever to say about whether the physical world is all that there is. It is this obvious, logical distinction between science and scientism/materialism that the ID movement fails to grasp in its entirety.
Nearly all of the hundreds of thousands of Christians who are practicing scientists find that evolution is no threat to their faith. The Biologos organization, founded by geneticist Francis Collins (now head of the Centers for Disease Control), helps evangelicals deal with questions like, “Why should Christians consider evolutionary creation?”. Evangelist Billy Graham put it well: “I believe that God created man, and whether it came by an evolutionary process and at a certain point He took this person or being and made him a living soul or not, does not change the fact that God did create man… Whichever way God did it makes no difference as to what man is and man’s relationship to God.”
Some General Tactics of Anti-Evolutionists
A core agenda of opponents of evolution is to try to cast doubt on the results and conclusions of mainstream science. One tactic, which we will see below in the case of whale fossils, is to reach back to some outdated studies which were based on preliminary evidence, and which have since been corrected by additional finds. Instead of celebrating these instances of the self-correcting nature of science, creationists use these cases to claim that scientists are always just making stuff up so you can’t believe any of it.
Because the fossil discoveries of the past 25 years have done so much to fill in the gaps on the fossil record, another tactic of evolution deniers is to reach back into the 1980’s or earlier and pull out quotes from actual scientists which seem to be admissions of the failure of the fossils to support evolution. But the only reason these quotes seem to oppose evolution is because the anti-evolutionists have taken them out of context and edited or spun them in a way quite opposite to the author’s actual meaning.
A few examples will suffice. A favorite quote is from Colin Patterson, then senior paleontologist at the British Museum of Natural History. In response to a letter from a creationist asking why he did not include direct illustrations of transitional fossils in his book, Patterson wrote back in 1979:
I fully agree with your comments on the lack of direct illustration of evolutionary transitions in my book. If I knew of any, fossil or living, I would certainly have included them…. I will lay it on the line—there is not one such fossil for which one could make a watertight argument.
This has been spun to be an admission that the fossil record does not support evolution. But that is not at all what Patterson meant.
The TalkOrigins site has a whole section dedicated to unmasking misleading creationist quote. This Quote Mine Project should be consulted whenever a seemingly damaging quote from a practicing scientist is cited by a creationist (a list of quotes arranged by author is here). For instance, the clarification there on the Colin Patterson quote points out several things. First, from Patterson’s book itself, it is clear that he acknowledges that there are series of fossils which bridge wide gaps between existing types:
“In mammals, for example, the gap between horses, asses and zebras (genus Equus) and their closest living relatives, the rhinoceroses and tapirs, is filled by an extensive series of fossils extending back sixty-million years to a small animal, Hyracotherium, which can only be distinguished from the rhinoceros-tapir group by one or two horse-like details of the skull.”
Second, if the entire text of Patterson’s letter is shown, instead of carefully edited excerpts, it is clear that his point was not to deny the existence of fossil series with gradually changing intermediate features. Rather, he was pointing out that for any particular fossil, it is impossible to say for sure (“make a watertight argument”) whether it is a direct ancestor or merely a side-branch “cousin”. But that is an obvious point, which we have already emphasized: the actual fossils found are almost certainly side-branch relatives rather than being on the direct lineage path.
Creationists also cite a number of quotes from Stephen Jay Gould from around 1980, such as:
The extreme rarity of transitional forms in the fossil record persists as the trade secret of paleontology.
and
The fossil record with its abrupt transitions offers no support for gradual change…All paleontologists know that the fossil record contains precious little in the way of intermediate forms; transitions between major groups are characteristically abrupt.
Again, the Quote Mine Project shows from Gould’s own writings that he was simply noting what we have noted above – – that evolutionary change is likely to occur relatively quickly (in geological terms) in occasional small isolated populations whose fossils will be so few that they will likely never be found. It is only the later, larger, successful populations who become established as a new genus or family that leave enough fossils to be found by us millions of years later. Thus, as Gould notes, the fossil record is expected to appear “jerky” rather than smoothly continuous at the species or genus level, but it does show transitional forms between larger groupings:
I count myself among the evolutionists who argue for a jerky, or episodic, rather than a smoothly gradual, pace of change. In 1972 my colleague Niles Eldredge and I developed the theory of punctuated equilibrium. We argued that two outstanding facts of the fossil record — geologically “sudden” origin of new species and failure to change thereafter (stasis) — reflect the predictions of evolutionary theory, not the imperfections of the fossil record. In most theories, small isolated populations are the source of new species, and the process of speciation takes thousands or tens of thousands of years. This amount of time, so long when measured against our lives, is a geological microsecond . . .
Since we proposed punctuated equilibria to explain trends, it is infuriating to be quoted again and again by creationists — whether through design or stupidity, I do not know — as admitting that the fossil record includes no transitional forms. Transitional forms are generally lacking at the species level, but they are abundant between larger groups. [From Stephen Jay Gould, bold emphases added]
The takeaway here is that whenever creationists cite some quote from scientists which seems to admit some fatal flaw in evolution, it should be assumed that this quote is out of date or out of context or misconstrued, unless the reader has traced it back to understand the full original meaning of the quote.
Some Objections Regarding Whale Fossils
Because whales are such a clear example of evolution, both ID and YE creationist spokesmen publish ongoing attacks on the evolutionary origin of whales. Every one of their objections, when fully assessed, is found to be lacking in logic or factuality, but to try to address all of these claims is tedious and unrewarding.
An opponent of evolution can put forth a plausible-seeming claim using just a sentence or two, but it may take several pages to present all the relevant background knowledge to show clearly why that claim is wrong. And my experience has shown that even if a scientist goes to all that trouble, nearly all creationists will ignore his response anyway. They will keep repeating the same (debunked) anti-evolution claim, or simply move on to the next and the next, until the scientist drops from exhaustion. For example, a year ago I had a written dialog with a YE creationist, where I painstakingly marshalled the facts which showed that the scientific claims in her slides were clearly incorrect. Although she was an intelligent, good-willed person, she simply waved away all these rebuttals.
All I can do here is take a random sampling of these objections and show why they fail. The reader who is interested in the truth on some other anti-evolution claim can use the information in this essay and do their own internet search to get to the bottom of the matter.
Regarding the fossils, here is a representative article by YE creationist Don Batten which tries to undermine the scientific evidence: Whale evolution fraud – Another evolutionary icon bites the dust
The main tactic here is to point out (and sometimes misrepresent) differences between some early proposals which were based on fragmentary preliminary finds, and later understandings after more complete fossils were available. The article claims that these differences destroy the value of these fossils as intermediates, but that is nonsense. The more recent and more complete skeletal features actually fit more readily into an evolutionary progression than the initial proposals did.
The first fossil finds of Pakicetus, published in Science by Philip Gingerich et al. in 1983, comprised only a piece of the skull and a piece of the lower jaw. The skull fragment happened to include the ear bones. The ear bones caused a sensation, since they had features we described above which is characteristic of whales and unique to them. These features are the thickened inner wall (involucrum) of the tympanic bone, and a wavy crest of bone (sigmoidal process). The jaw showed Pakicetus to be a carnivore, like modern whales. Analysis of the ear indicated that it was better suited to hearing in air than under water. With that information to go on, it was reasonable to suppose the animal as a whole would look much like a modern whale, except with legs to allow it to crawl out of the water and into the air. Although the 1983 technical paper confined itself to sober analysis of the fossils themselves, the cover of that issue of Science sported a speculative painting of the entire animal. It looked like a whale or seal with stubby legs or flippers, maneuvering underwater and closing in on a hapless school of fish. That was perhaps entrepreneurial overreach by Gingerich, as was his supplying whole reconstructed skulls to various museums, based on the two fairly small fragments which had actually been discovered. YE creationists claim that Gingerich made these skulls with bogus “blowholes”, but that is hardly accurate; the blowholes on modern whales are all the way back, on top of the skull itself, whereas the Gingerich’s proposed Pakicetus skulls just have the nostrils set a little way back from the tip of the snout.
Thewissen et al. published in 2001 on the finding of a more complete Pakicetus skeleton. From these remains, it was clear that although Pakicetus was adapted for hunting for fish in rivers, it was basically a terrestrial carnivore. It had long legs, and looked more like a wolf than a modern whale. The nostrils were at the tip of the snout, not set back. Considering that Pakicetus fossils date back to about 55 mya, this terrestrial morphology actually fits in better with the series of other cetacean fossil intermediates than if (as depicted in Science in 1983) Pakicetus resembled a modern seal. This is an example of the self-correcting nature of modern science, and a firm support for evolution.
So (back to the Batten article) what can the YE creationists complain about here? Well, they complain that some of Gingerich’s original skulls were still on display in the American Museum of Natural History (AMNH) and a few other museums as late as 2014. That doesn’t change the facts about Pakicetus which make it a plausible whale precursor. On the AMNH website we find this accurate image, which dates back to 2006…

…along with the accurate text, “Straddling the two worlds of land and sea, the wolf-sized animal was a meat eater that sometimes ate fish, according to chemical evidence. Pakicetus also exhibited characteristics of its anatomy that link it to modern cetaceans, a group made up of whales, porpoises, and dolphins.” YE creationists scoff at classifying Pakicetus as a cetacean, but the fact is that the involucrum of the earbone is considered to be a defining characteristic of cetaceans, and Pakicetus has that feature.
So the YE creationist case against Pakicetus simply falls apart upon inspection. The same is true for its attacks on Ambulocetus and Rodhocetus – – there are complaints that some preliminary proposals on their anatomy based on early fossil finds turned out to be incorrect (e.g. proposed flippers or tail fluke), and that some museums have been slow to update their displays. These are all just red herring distractions. Nothing here obviates the intermediate features of the actual, more fully known fossils of these animals. The image we displayed above for Rodhocetus, with correct legs (not flippers) and correct tail (no fluke) dates back to the 2001 cover of Science. So as with Pakicetus, the corrected understanding of these fossils has been publicized for at least twenty years now.
A “gotcha” video interview with Hans Thewissen embedded in the Batten article tried to make it seem like evolutionists have sneakily inserted a “blowhole” in their reconstructions of Ambulocetus, but this is misrepresentation. Again, a “blowhole” implies a hole on top of the skull, not nostrils in the snout. Fossils of Ambulocetus do not include the snout, so we simply don’t know where the nostrils were located. Since the morphology of Ambulocetus lies in general between Pakicetus (where the nostrils are in the tip of the snout) and Rodhocetus (where the nostrils are set back significantly from the tip), it is reasonable to depict Ambulocetus with nostrils at the front (compared to today’s whales), but set back a little from the tip. This is exactly what the reconstructions show. Since there is no direct fossil proof of where exactly the nostrils were located, scientists do not make this position a part of their arguments for evolution. Thus, this issue is just another deceptive distractor by YE creationists.
Finally, another video segment with Thewissen deals with features of the ear of Ambulocetus. The caption on the video claims that the case for including Ambulocetus in whale ancestry “rested mostly on this ridge of the ear”, pointing to the sigmoidal process on the edge of the tympanic bone. The video interviewer then leads Thewissen (who is not a native speaker of English) into acknowledging that this ridge in Ambulocetus is “as questionable” as the ridge in Pakicetus (“Gotcha!”). At that point the sound in the video suddenly cuts out, even though visually Thewissen is continuing to point to that ridge and presumably continuing to comment on it.
Let’s analyze this video. First, it is not true that the case for Ambulocetus rests “mostly on this ridge of the ear”. The case for ancestry involves the whole complex of features of this fossil, with mixed terrestrial and aquatic adaptations and being found in the appropriate timeframe. Second, as Thewissen notes in the video, the Ambulocetus ear fossil has been heavily crushed in the fossilization process, such that the thickened involucrum (which is even more of a characteristic feature of cetaceans than the sigmoid process) is not fully accessible, though it is present; the more delicate features of the thinner sigmoidal process may well have been partly mashed out. Third, as noted above, the Pakicetus sigmoidal process is indeed somewhat different (more plate-like) than seen in modern whales, but it is not surprising to see some differences. Pakicetus is acknowledged to live mainly on land, where hearing in air is advantageous, whereas modern whales live entirely underwater, and have sound conducted to their ears mainly via their jaw bones. For the Ambulocetus ear to be classed with Pakicetus in this regard is not a problem, especially considering Ambulocetus lived millions of years before modern whales. Finally, it is suspicious that Thewissen’s voice is cut off at the 2:40 mark as he is continuing to point at the sigmoidal process, such that we are denied hearing what further clarification he might be offering. The video is also edited at the 2:19 mark to remove whatever other comments Thewissen made when he first introduced the sigmoidal process. At any rate, Thewissen and nearly all other fossil experts who have examined the Ambulocetus ear are satisfied that it does display the key involucrum and also a reasonable representation of the sigmoidal process.
All this discussion of one short YE creationist article illustrates how tedious it can be to refute anti-evolution claims. We will briefly consider two more fossil-related objections.
Opponents of evolution have objected that the tiny hind leg appendages of the basilosaurids might have had some useful function such as clasping or positioning during mating, and therefore they are not vestigial legs. This objection totally fails. First, whether or not a vestigial structure acquires some other function has no bearing on whether or not it is a vestigial item. The structure simply is that of vestigial legs: similar detailed physical features as terrestrial legs (see figure below), but shrunken way down in size, as evolution would predict for a fossil whale intermediate. Second, even for fully terrestrial animals, hind legs can have a function in mating. If you amputate the hind legs from a pair of dogs, you will get no puppies. So even if these tiny appendages were useful in mating, that would display continuity, not discontinuity, with earlier cetacean ancestors. Finally, we don’t know whether these appendages were actually useful or not in mating; that is simply speculation. Modern whales get along fine mating without any hind legs at all.
A Basilosaurus hindlimb is shown below. Although this limb is far too small to support any of this animal’s weight, it has the features of a typical terrestrial mammal hindlimb, such as femur (thighbone), knee joint with patella (kneecap), fibula and tibia in calf, ankle joint and footbones (metatarsals and phalanges). Common ancestry via evolution predicts this, whereas ID and YE creationism do not. [2]
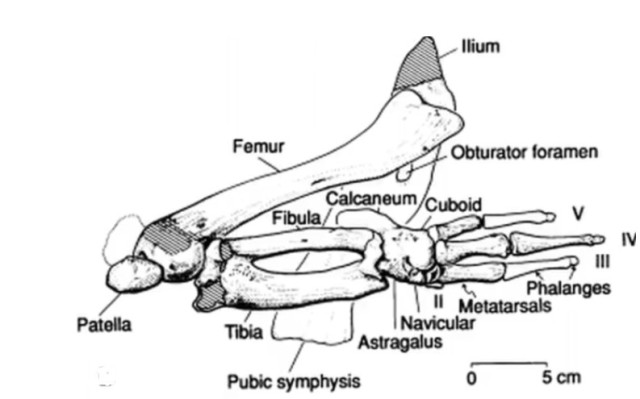
Hindlimb of Basilosaurus isis. Source: Gingerich, et al., Science (1990), Vol. 249, pp. 154-157
Let us look at the forelimbs of whales as well. They look from the outside rather similar to shark forelimbs, which are made of bone and cartilage. Wouldn’t it be economical of an Intelligent Designer to use the same shark-like bone/cartilage structure in whale forelimbs? However, as this figure shows, whale forelimbs have the same bones as the forelimbs/arms as terrestrial mammals such as humans and cats. YE creationism and ID would not predict this, but evolution does.
One last fossil dispute considered here involves a basilosaurid-type fossil discovered in Antarctica. This issue is discussed at length by paleontologist (and evangelical Christian) Ryan Bebej. A partial jawbone with fragmentary teeth, which appeared to come from a basilosaurid, was found in a rock layer in Antarctica. This fossil was initially dated as being 49.5 million years old, which would be older than previously discovered Basilosaurus fossils (34-41 mya). Is this a problem for evolution? No, for several reasons. First, while the fragmentary jawbone does resemble known basilosaurid fossils, we know from the controversy over early Pakicetus finds that we should not conclude too much from just a few fragments. We simply don’t know what the rest of this Antarctic fossil creature looked like.
Second, we never find every fossil from every individual from every species. Therefore, whatever date range is currently known for the fossils of a given species or family, it is nearly certain that that species or family actually existed before and after that range. In paleontology this is known as the Signor-Lipps effect: “Since the fossil record of organisms is never complete, neither the first nor the last organism in a given taxon will be recorded as a fossil.” If basilosaurids were already known to exist by 41 mya, it is quite possible that some early form of them appeared several million years earlier. Likewise, although Rodhocetus fossils are only known from about 46-47 mya, there were almost certainly Rodhocetus populations living well before and after this narrow range, although we have not yet found their remains. The same holds for all the whale precursors, including Pakicetus and Ambulocetus.
It is also worth repeating that paleontologists do not claim that basilosaurids descended directly from Rodhocetus or the other known fossil species with intermediate features. Whatever the direct lineage to basilosaurids is, it likely split off before the appearance of Ambulocetus and split off before the appearance of Rodhocetus. As we have stressed repeatedly, the species which form large enough populations to leave an appreciable fossil record are nearly all side-branches on the evolutionary tree, not direct ancestors.
Third, it turns out that those particular Antarctic rock layers are intrinsically difficult to date. Other studies, based on more recent data, date this rock layer to be significantly younger than 49 mya. In his article on the Biologos website, Bebej discusses a series of videos produced by the ID organization Discovery Institute which argue for the older date. He finds overall tone of the videos to be “disheartening” since, “The manner in which these videos are produced often reduces serious scientific claims to little more than silly jokes and analogies.”
On the technical content, he finds, “These videos are rife with inaccurate portrayals and misconceptions about the fossil record, as well as technical subfields like biostratigraphy and phylogenetic systematics.” Regarding the geostratigraphic evidence for the rock layers, Bebej notes:
These videos do a poor job of fairly presenting the various scientific claims about the disputed age of these layers. They make it seem like most reliable studies prefer the older dates, with just a few rogue unreliable studies positing younger dates. But in reality, there have been many studies over the years that have favored the younger ages for these units for a variety of different reasons.
Some Genetics Claims by Opponents of Whale Evolution
The genetics of whale evolution is complex technical subject area, which we will not try to unravel here, beyond a few general comments. While some of the genetic trends discussed here are straightforward, such as disabling or down-regulating mutations in genes for hind legs and tooth enamel in major classes of whales, it should be noted that it is often more difficult to determine the exact phylogenetic relations among individual whale species and families. Known factors such as incomplete lineage sorting, convergent evolution, interbreeding of closely related species, and other gene flows can cause different study approaches to give different phylogenies.
A persistent claim by ID proponents (who accept the geological dating of the various transitional cetacean fossils, including those in the 30-60 million year time frame) is that there was “not enough time” for the needed mutations to occur by natural processes. In fact, they argue, that practically any kind of evolution for any large mammal is essentially impossible: the time to achieve a mere four pre-specified mutations for humans is [they claim] about six billion years, which is longer than the earth has existed. In the case of whales there are dozens of mutations involved, so whale evolution is alleged to be utterly unthinkable.
You can easily show that this argument is bogus by taking yourself as Exhibit A. Your genome has about fifty mutations in it, relative to your parents. These are outright mutations, in addition to the usual recombination of chromosomes that happens with sexual reproduction. According to the ID logic, it should take trillions upon trillions of years for your particular genome to appear, so it is essentially impossible that you exist. But here you are, created from your parents in a single generation.
There is obviously a big fallacy in the ID logic here. What’s the catch? The mistake is aiming for some particular pre-specified set of mutations. If, before you were conceived, someone were supplied with a printout of a set of 50 pre-specified mutations placed in pre-specified locations amongst 3.1 billion base pairs of human DNA, they would rightly calculate that the odds to be infinitesimally small of this specific genome appearing in one of your parents’ children. And yet, here you are!
The underlying, unspoken false premise is that there is only one possible specific genome for a whale, or a human being, and so evolution is constrained to aim at that very narrow target. The reality is that there are billions and billions of possible sets of mutations which will give a viable whale or human genome; some mutations will enhance fitness (and thus be favored by natural selection) and some will not. So the notion of aiming at some particular pre-specified set of mutations is nonsense, and thus the whole ID “not enough time” argument falls apart. This fallacy has been pointed out to the ID folks at the Discovery Institute over and over again, but they simply ignore it since it destroys their case. [3] [4]
A 2018 dialog at the Biologos site covered various issues around the “not enough time for whale evolution” claim. There are a number of informative comments and links there. A more extensive dialog on the Peaceful Science site treats this subject as well. Two entries from that dialog are reproduced below. The first comment notes that many (not all) of mutations involved in going from terrestrial animals to fully aquatic whales were fairly “easy” ones like just deactivating or down-regulation some existing genes, rather than developing whole new genes [5]. This goes a long way toward explaining how this evolutionary transition was accomplished relatively quickly:
It is remarkable how many of the changes required for whale evolution are caused by loss of function mutations (which end causing “pseudogenes”), or small tweaks to proteins. This is one of the big surprises of mammalian evolution. Large changes can take place with tweaks to the genetic code. Eyes adapt to underwater vision by losing a rhodopsin gene. Hind Limbs are lost with the loss of a homeobox gene. Taste buds are lost when two genes are lost. Smell receptors are almost entirely lost in most species too. In all these cases, we see remnants of the broken genes, and in many cases the details of how these losses increase function are well understood.
Remarkably, it does not appear any new enzymes or de novo genes are required in whale evolution. It appears that small tweaks to existing proteins, or loss or alteration of the function of existing genes, account for the changes we see at this point.
Also, there does not appear to be any reason that a large number of these changes must happen at the same time. They appear gradually in the tree, and it’s not clear at all why they would need to be “coordinated”. They do not appear to need to occur at the same place and time to be useful. So this does not make these transitions unlikely.
In response to a question as to why we haven’t observed many dramatic evolutionary changes in the span of recorded human history, it is noted that (a) human history time span (~10,000 years) is tiny relative to the geologic ages (millions of years) for evolution, and (b) the starting point for whale evolution (e.g. pakicetids or their cousins) already had some whale-like characteristics:
In terms of changes, that has been merely 10,000 years, and we do not expect much. However, we have seen very large changes. Most clearly in domesticated animals. For example look at dog and plant breeds. Cabbage, broccoli, kale, brussel sprouts, and more all come from the same plant that was selectively breeded.
In terms of intermediate forms that parallel whale evolution, there are several animals on the continuum. Sea lions, seals, walruses, hippos, beavers, otters…they all have features that make them look like they are half way between a land animal and eventually becoming something like a whale (if it wasn’t that whales were already occupying that niche). The have several parts the intermediate features that are supposedly required to evolve in one fell swoop. For example, Hippos have internal testes, but seals have semi-external testes with no scrotum. If whales were to all die off, and there a large number of sea niches available, who knows, maybe in 4 million years there would be seal-like whales in the ocean.
Also, it is important to keep in perspective what we mean by “whales” appeared in just 8 million years. Those whales were very different than the whales we see today. There were no blue whales, for example, because baleen had not appeared yet. It is not as if these early whales were the same as whales we see today. I am pretty sure that some of them, for example, had external legs still.
Moreover, the starting point was not a prototypical land animal (e.g. a dog), but something closer to a carnivorous hippo, that had a very whale like head. So it is not as far of a leap as you are envisioning. A better analogy would be, as I put forward a moment ago, a seal evolving into a more aquatic form.
…Hippos and whales are from the same line. I’m not sure we precisely know the ancestral state. For example, it is possible (I’m not sure we know) pakicetus (the whale-headed rat dog) had internal testes [since both hippos and modern whales have internal testes]. In that case, going from pakicetus to a whale would not require a change in this trait.
Readers here who have issues around the genetics of whale evolution should take their concerns to Biologos or Peaceful Science, where experts are standing by to answer your questions. It looks like the two dialogs referenced above are still open.
CONCLUSIONS
We started by posing some of the challenges to evolution presented by whales. Whales have key features marking them as mammals, and mammals started off as terrestrial animals. If whales became whales by way of evolution, then there are well defined features which we should find in their genomes and in the fossil record. Even though modern whales normally have no hind legs, evolution predicts that they should still have the genes for making hind legs, with the genes in some deactivated or down-regulated state. Likewise, we should find deactivated genes for making teeth in the baleen whales which have no teeth today.
Modern gene sequencing technology does show that all these features predicted by evolution are in fact present. We also find other deactivated genes, such as some of those for smelling and for blood clotting, which correspond to reduced olfactory sensing and reduced propensity for undesired clotting while diving. These gene deactivations go a long way towards explaining how modern whales evolved relatively quickly from terrestrial animals: it is much easier to deactivate or down-regulate existing genes (in order to achieve a change in body shape and function) than to evolve whole new genes.
Typical terrestrial mammals have nostrils at the front of their snouts. All modern whales, however, have nostril openings or blowholes all the way back on the tops of their heads. If evolution is true, we expect to find various fossil intermediate species with nostrils at midway points on the snout. Similarly, we expect to find fossil intermediates with regular terrestrial hind legs for the earliest specimens, and then stubbier and stubbier legs and finally no external hind legs at all. The fossil record does show all these features, as predicted by evolution.
In sum, whale origins from terrestrial ancestors puts the theory of evolution to a definite challenge, and this challenge is plainly met:
Genes for hind legs and teeth are there in all modern whale genomes, as predicted by evolution.
Fossils with ranges of intermediate features (leg sizes, nostril positions, etc.) have been found, as predicted by evolution.
This is how genuine science progresses over time: testable hypotheses are proposed and then subjected to real-world evaluation. Proposals which fail their tests are either modified or rejected, while those that pass their tests are provisionally retained. In contrast, young earth creationism and Intelligent Design do not provide these sorts of detailed, falsifiable predictions for whale origins, and thus do not qualify as science.
As an evangelical Christian, I used to worry that evolution somehow invalidated the Bible. I have come to believe that it does not. The core Christian truth claims concerning the life and work of Jesus Christ stand on their own, well-supported by historical evidences (see Historicity of Jesus ). The Genesis creation narrative was not written to provide a physical explanation of geology to 21st century readers. Rather, it provided the ancient Hebrew people a conceptual framework to understand that there was only one God, who was in control of nature and who had good purposes for humankind, in contrast with the religious views of the surrounding peoples with their capricious and often cruel gods. To demand that Genesis present a natural history in modern scientific terms is to simply make a category mistake.
FURTHER RESOURCES
Many links appear in the text to articles on whale evolution which are available online. For a short introduction to these issues, see the two-page Berkeley Whale Evogram.
For a brief video presentation of evidence for whale evolution, see the middle part of this 11 minute YouTube: What is the Evidence for Evolution?
The Tale of a Whale is a 24 minute video by Biologos which goes into more depth on the evidence for whale evolution.
ENDNOTES
[1] The mainstream Discovery Institute ID effort is quite evasive about providing a positive model of how the Intelligent Designer actually did anything. As noted, however, “Intelligent Design” is a big tent. Hugh Ross’s Reasons to Believe ministry would fall into the larger ID camp (old earth but anti-evolution), but his organization does propose a model of biological origins which is intended to be testable.
[2] After the fact, ID or YE creationism could always say of some biological feature, “Well, that is just how God/The Intelligent Designer chose to do it.” That statement is true enough, but (1) it shows that ID does not qualify as science, since there is nothing which could, even in principle, test its validity, and (2) it raises the question of why the Intelligent Designer would make things, again and again, which are exactly what evolution predicts. Surely there are other ways to make a stubby appendage in a marine animal than to make it an exact replica of a terrestrial animal leg.
[3] ID proponents sometimes point to 2004 experiments by Discovery Institute research Douglas Axe as supporting their contention that evolution is severely hampered by having only very narrow “targets” to aim at. However, his experiments were largely irrelevant to actual evolving systems. Nick Matzke pithily summed up the significance of Axe’s work: It “typically involves making evolutionarily absurd modifications to proteins and then showing that they don’t work.” For a more thorough critique of Axe’s experiments, see this essay by Arthur Hunt. For an assessment of more recent claims by Axe, see this 2018 Biologos discussion and links therein.
[4] Contrary to ID’s claims, we can actually observe in the lab the appearance of multiple mutations that give new functions. See Evolution Before Our Eyes: Complex Mutations in Microbes Giving New Functions. One of the examples in this link involved a binding site which involved four mutations to bind a new protein. This refutes the claim of ID researcher Michael Behe that the appearance of a new protein binding site which requires more than two mutations is, for all practical purposes, impossible – – beyond the “edge of evolution”.
[5] The evolution of whales largely by loss of gene functions naturally raises the question of whether all evolution is like that and where did all the starting functional genes come from. Just busting genes does not seem like a great overall strategy for evolutionary progress. Two comments may be made here. First, the same kinds of genetic comparisons which show that some older genes have been “broken” over time also indicate that other, new functional genes have been evolved over time. Studies indicate that in vertebrate lineages around 25-30 new genes appear every million years (with great variation among different groups) via mechanisms such as duplication and de novo generation. Thus in many lineages (perhaps not whales), the total number of functional genes may be relatively constant, with new genes somewhat balancing the loss of earlier ones.
New genes do not appear out of thin air. Rather, they are formed by modification of existing DNA. Mechanisms for modification include duplication, deletion, fusion, fission, rearranging pieces such as exons, and so on. All these mutational modes can be observed to operate in organisms in the lab, here and now. Discerning exactly how these modifications impacted the lineage of a particular species or group requires careful comparisons of genomes among various families. The figure below from a 2013 review by Long, et al. illustrates some of these known mutational mechanisms with specific examples of new genes formed by these mechanisms.

Source: M Long, N. W. VanKuren, S. Chen, and M. D. Vibranovski, “New Gene Evolution: Little Did We Know”, Annual Review of Genetics, Vol. 47:307-333 (2013) [As with all images here, click it to see clearer image]
Caption for this figure above (supporting references deleted):
“Representative new genes exhibiting various new gene origination mechanisms. A) Jingwei, a new gene found only in D. teissieri and D. yakuba, was generated by a combination of retroposition, DNA-based duplication and gene recombination which formed a chimeric gene consisting of Adh-derived enzymatic domain and a hydrophobic domain from Ymp. B) PIPSL in humans is a consequence of gene fusion between two adjacent ancestral genes by read-through transcription and subsequent co-retroposition. C) Gene fission split the ancestral gene monkeyking into two distinct genes in D. mauritiana, revealing an intermediate process of gene fission aided by gene duplication and complementary degeneration. D) The gene ENSMUSG00000078384 in mouse revealed the evolutionary process of de novo gene origination . E) Two new genes in humans, DAF and mNSCI, were generated by domesticating transposable elements, Alu and short interspersed elements (B1-B4) . DAF and Alu elements is a neat case where alternative splicing generated a new isoform in the mammalian genome. F) Horizontal gene transfer (HGT) is prevalent in bacteria with mechanisms including homologous recombination. Antibiotic resistance genes can be acquired by host genomes containing the intl gene, which encodes integrase, a recombination site (att), and a promoter to express the captured gene, as depicted by the process on the left.”
A second comment regarding evolution via gene deactivation is that, yes, it is possible that this process could lead in some cases to a gene-depleted species which is hyper-specialized for a particular environment, such that this species does not have the genetic diversity to evolve in some other direction if the environment should change. It is sobering to note that scientists are convinced by various lines of evidence that more than 99% of all species that have existed on earth have gone extinct.

Since whale evolution is a controversial topic, here is the Comments Policy as posted in About:
“There is a quick registration for leaving comments (just asks for a username and e-mail address). Comments are expected to be reasonably courteous and to reflect the commenter’s own thoughts or questions (no links to other sites or videos), and to relate directly to the material in the blog post. Any factual corrections are welcome, while comments with extraneous claims will likely not be published.”
(I try to do readers the courtesy of responding to most comments, and have learned over time that it is not a fruitful use of my time to interact with someone who just wants to repeat various anti-evolution claims that have been already refuted over and over again).
As noted in the post, if a reader wants to dispute genetic issues, he or she should take their concerns to the referenced Biologos or Peaceful Science forums, where experts can answer their questions.
This is a very detailed and thorough compilation of facts, which will make a good reference for years to come.
Thanks, Jim. Hope you are weathering this last blast of covid safely…
ISTR that there was a review (I think in Nature, 2000), still quoted by creationists, about disagreements between palaeontologists and molecular biologists in constructing phylogenies. The whale was a prize example. However, finding the astralagus in, I think, Pakicetus was enough to convince the palaeontologists that the molecular biology phylogeny was correct.
Do you have any details on this story? It illustrates several things; the self-correcting nature of science, the importance of anomalies in advancing knowledge, and the obtuseness with which absolutists regard anomalies as refutations
I tried to cover this issue briefly with ” …The closest genetic relative to today’s whales is the hippopotamus. In fact, the closest genetic relative of the hippopotamus is not some other land animal, but the whales. This relation was controversial when it was first discovered, because whales are carnivores, whereas hippos are primarily herbivores. Prior to various fossil finds around twenty years ago, an extinct group of carnivores called mesonychids was widely proposed as terrestrial ancestors for whales.
Hippopotamus is a type of artiodactyl. Artiodactyls are hoofed animals (ungulates) with an even number of weight-bearing toes. A distinctive skeletal feature of all living artiodactyls is an ankle bone (astragalus or talus) …”
As of 1990s, the genetics crowd was claiming hippo (artiodactyl) as nearest relative of whales. That did not sit well with the paleontologists since hippos are mainly vegan; the fossil boys had long touted mesonychids as whale ancestors. So, yes, there was disagreement back then, but as you stated, the anklebones of Pakicetus etc have resolved this to nearly everyone’s satisfaction. Which, of course, does not stop the YECs from citing the old, outdated disagreement…
Agreed on all counts, and I did not wish to suggest that you had missed this in your excellent post.
The reference I’m thinking of would be useful in two ways; it would give quite a detailed history of the disagreement that you describe, and a web search on it would give links to creationists continuing to use it. The latter is something I would find useful in my own writing about them. If I come across it, I will send it to you, and hope you will do likewise.
I appreciate your bringing this up. I have tried to anticipate objections, at least some of them, and preemptively address them in the text. I, too, have a dim memory of claims being thrown about of contradictions between fossils and genetics re mesonychids, referring back to c. 2000 era. But I just spent an hour searching the web, and have not found much in the way of clear reference to this in current YEC articles. Maybe most of them have tiptoed away from this claim, now that the controversy has been resolved?
Probably somebody somewhere will keep claiming contradictions, regardless of the facts.
Anyway, I’ll tweak this a little more to try to clarify why this was an issue but is not any more.
And, as I noted elsewhere in the article, trying to do real world phylogeny is tricky — – there are various genuine factors (convergent evolution, incomplete lineage sorting, etc) that make it hard to disentangle all the threads, so there will always be cases where you can focus on one set of evidence and get a different result than if focus on another. So, sadly, there will always be fodder for them to try to spin.
If the creationists are now steering away from this, perhaps it’s because because even the disagreements show more agreement than they can stomach
Well, I finally found a creationist site citing the old 1990s controversy — citing a 1997 article :
Shimamura et al., Nature 388 (1997), pages 666 -667, “Molecular evidence from retroposons that whales form a clade within even-toed ungulates” (Ev)
HERE IS LINK TO THIS 1997 NATURE ARTICLE: https://www.nature.com/articles/41759
Caption for their Figure 1 – – The two newly isolated families of SINEs, CHR-1 and CHR-2, were present exclusively in the genomes of whales, ruminants and hippopotamuses.
…and text which called out the apparent dilemma:
The conclusions from our retropositional analysis are inconsistent with earlier morphologically based hypotheses16,20,21. Paleontological and morphological data suggest that modern whales originated from the Archaeocetes (primitive aquatic cetaceans), which first appeared in the early Eocene epoch22. The Archaeocetes are believed to have originated from mesonychians, which appeared before the Eocene20….
Again, all this is resolved with the further fossils
Thanks, Scott. exactly what I was looking for. And since it was cited over 300 times, I can get any more detail I need through the Web of Science Citation index
AiG posted this in September last year: https://answersingenesis.org/aquatic-animals/isnt-the-whale-transitional-series-a-perfect-example-of-evolution/
Legs too long, or too short, or pictures are based on reconstructions, or the skeletons are too similar or too dissimilar to modern whales or to land tetrapods, and that sort of thing (by someone called Troy Lacey, who doesn’t seem to have a bio, but does seem to have done his homework at least from some review or textbook account or something). Not a mention of the molecular evidence, despite a sentence that reads “And what of new genetic claims that hippos share more common DNA with whales?”
It has taken me a long time to come around to this view, but I now believe that writers such as this are being consciously dishonest, rather than merely perverse, in their selection of data. Whether they are being honest about anything is a more difficult question.
As it happens, I had recently committed to giving a talk to a student group in Texas for which I had chosen the title “from Jonah to Genesis, by way of Pacicetus”. Synopsis: Jonah (and I know it was a big fish) was never intended to be regarded as little truth, because of what the audience would know about Nineveh. Bulk of talk; summary of evidence for whale ancestry (the serological evidence was actually among that barred from presentation at the Scopes trial). Wrap up: Genesis has whales created before land animals. But we’ve just seen that they were descended *from* land animals. So *either* you reject all the science, *or* you accept that Genesis is to be understood allegorically, like Jonah is to be understood allegorically. Your moral choice.
So your article here has done half my work for me already. Thanks.
Paul, glad to hear some of this material can help you. As I have said, I respect your integrity and your tone in dealing with folks you disagree with.
re conscious dishonesty…it is really mindbending to contemplate what these folks are thinking. As I noted, they (for other reasons) *know* that whales did *not* evolve, so on one level it is proper for them to view the evidence through that lens.
But there are two approaches to this. Kurt Wise and Todd Wood treat the evidence accurately, acknowledging that it seems consistent with evolution, but say, “nevertheless, I still believe in special creation because that’s what the Bible teaches”.
Then there are the rest of the YE creationists…. I couldn’t do what they do. But I could never be an effective lawyer or salesman or politician, either, for the same reason. I guess folks get so deep into their views that “all’s fair in love and war”. And the general culture is getting pretty intolerant and uninterested in truth, compared to promoting the party line.
I am not sure what the audience is for whatever you are writing, but as you are aware the underlying driver for opponents to evolution is usually not the science (as witnessed by relatively few folks oppose evolution unless they have a religious reason). Interesting that is is mainly Protestants, while Catholics are OK with evolution, thanks to a series of popes who took openminded stances, per https://letterstocreationists.wordpress.com/2014/11/04/the-pope-speaks-on-creation-and-evolution/
Anyway, as my wife keeps reminding me, in any piece which might be read by opponents of modern science, it can be helpful to always include a few pointers as to how other conservative Christians interpret the Bible, though you may not a personal stake in this issue. Feel free to use or paraphrase anything I have written on this topic, I don’t care about being referenced.
I usually make reference to Billy Graham, since he has/had a lot of credibility in that camp. (Though there are some superfundamentalists who find him not conservative enough…)
Thanks. Good advice
Pingback: How Whales Got To Be Whales – Economist Writing Every Day
Pingback: Fazale Rana Disputes Evolution [2020 NCCA Apologetics Confc, 3] | Letters to Creationists
Pingback: John Sanford Disputes Evolution [2020 NCCA Apologetics Confc, 5] | Letters to Creationists
Pingback: Realistic Expectations for Transitional Fossils | Letters to Creationists
Pingback: Biologos and Peaceful Science Forums Address Timely Evolution/Creation Issues | Letters to Creationists
Pingback: Scientists’ Responses to Stephen Meyer’s Return of the God Hypothesis | Letters to Creationists
Pingback: Tracking Young Earth Creationist Organizations by Joel Duff at The Natural Historian | Letters to Creationists